 
Each chemical element has its own characteristic emission spectrum. Positions of the emission lines tell us which wavelengths of the radiation are emitted by the gas. This spectrum is seen as colorful lines on the black background (see Figure 6.15 and Figure 6.16). The emission spectrum is observed when light is emitted by a gas. The missing wavelengths tell us which wavelengths of the radiation are absorbed by the gas. This spectrum appears as black lines that occur only at certain wavelengths on the background of the continuous spectrum of white light ( Figure 6.13). An absorption spectrum is observed when light passes through a gas. The difference between the absorption spectrum and the emission spectrum is explained in Figure 6.14. During 1854–1861, Gustav Kirchhoff and Robert Bunsen discovered that for the various chemical elements, the line emission spectrum of an element exactly matches its line absorption spectrum. Solar absorption lines are called Fraunhofer lines after Joseph von Fraunhofer, who accurately measured their wavelengths. When we use a prism to analyze white light coming from the sun, several dark lines in the solar spectrum are observed ( Figure 6.13). 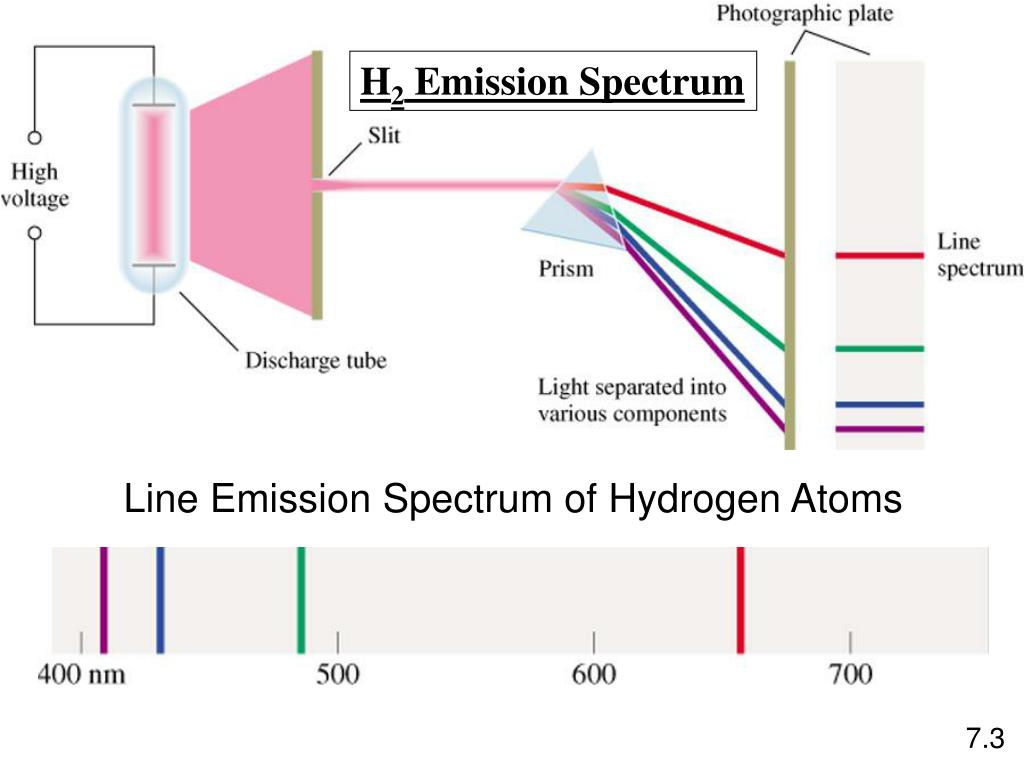
To understand the specifics of Bohr’s model, we must first review the nineteenth-century discoveries that prompted its formulation. The model has a special place in the history of physics because it introduced an early quantum theory, which brought about new developments in scientific thought and later culminated in the development of quantum mechanics. Historically, Bohr’s model of the hydrogen atom is the very first model of atomic structure that correctly explained the radiation spectra of atomic hydrogen. Summarize how Bohr’s quantum model of the hydrogen atom explains the radiation spectrum of atomic hydrogen.Describe the postulates of the early quantum theory for the hydrogen atom.Explain the atomic structure of hydrogen.Describe the Rutherford gold foil experiment and the discovery of the atomic nucleus.Explain the difference between the absorption spectrum and the emission spectrum of radiation emitted by atoms.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
